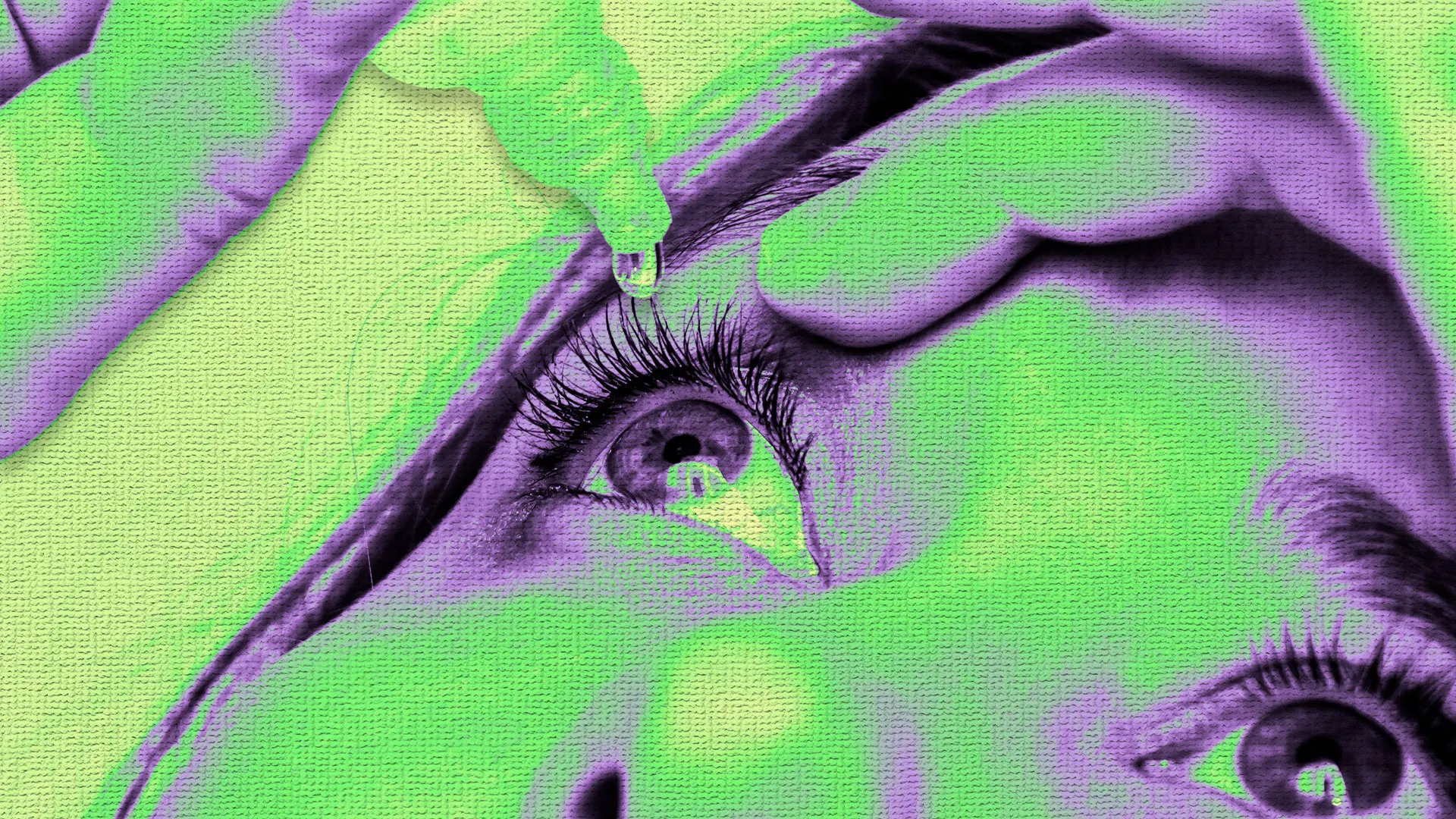
"Using nonsterile eye drops that harbor bacteria and fungus can cause eye infections, which can become severe because the immune system has a hard time accessing the eyeball and fighting the microbes."
"This recall is very large, potentially affecting over a million people. It is the second time since 2023 that the Food and Drug Administration has become aware of sterility issues at K.C. Pharmaceuticals."
K.C. Pharmaceuticals recalled more than 3.1 million bottles of lubricating eye drops due to inadequate sterility testing. This recall affects eight products sold under various names at major retailers. Using nonsterile eye drops can lead to severe eye infections. This is the second recall related to sterility issues at K.C. Pharmaceuticals since 2023. Consumers are advised to check their eye drop products purchased since April 2025 against the recall list on the FDA site for specific lot numbers and expiration dates.
Read at Fast Company
Unable to calculate read time
Collection
[
|
...
]