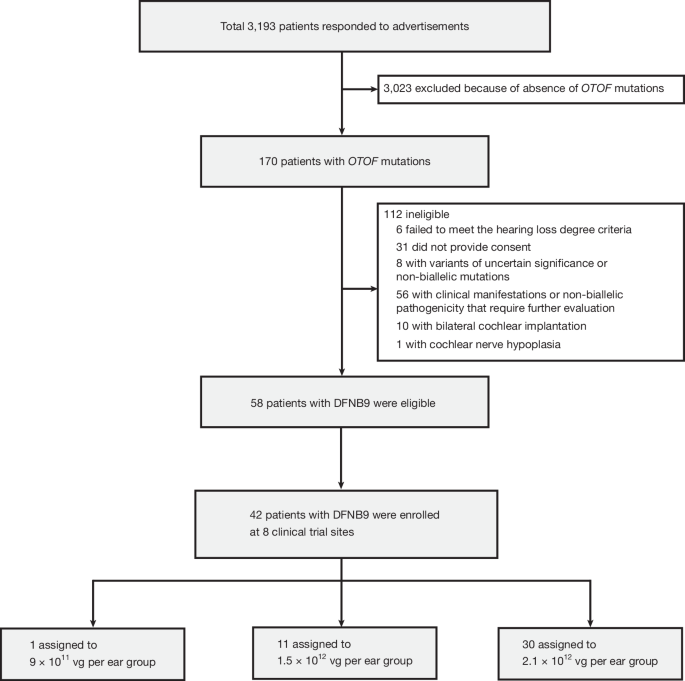
"In this single-arm, multicentre trial conducted at eight centres, 42 participants (aged 0.8-32.3 years) received adeno-associated virus (AAV) serotype 1 carrying a human OTOF coding transgene (AAV1-hOTOF) at three vector dose groups, with up to 2.5-year follow-up."
"The primary end point was dose-limiting toxicity within 6 weeks. The secondary end point assessed efficacy and adverse events. No dose-limiting toxicities were observed."
"Grade 3 adverse events included decreased neutrophil count. Hearing was assessed as part of the efficacy evaluation, indicating potential benefits of the therapy."
A multicenter trial evaluated the safety and efficacy of AAV1-hOTOF gene therapy for autosomal recessive deafness caused by OTOF gene mutations. Forty-two participants aged 0.8 to 32.3 years received the therapy in three dose groups, with a follow-up of up to 2.5 years. The primary endpoint focused on dose-limiting toxicity within six weeks, while the secondary endpoint assessed efficacy and adverse events. No dose-limiting toxicities were reported, although some participants experienced grade 3 adverse events, including decreased neutrophil count.
Read at www.nature.com
Unable to calculate read time
Collection
[
|
...
]