"The ability of cancer cells to adopt new phenotypes without further DNA mutations is now well understood to substantially influence tumour behaviour. Such plasticity has long been observed in melanoma, where early studies identified transcriptomic and phenotypic states not linked to specific genetic lesions1. More recent evidence indicates that most tumours encompass a heterogeneous yet reproducible number of transcriptional states3,4. The extent to which tumour cells transition between states is an open area of investigation and has been hypothesized to be regulated by cues from the tumour microenvironment (TME)."
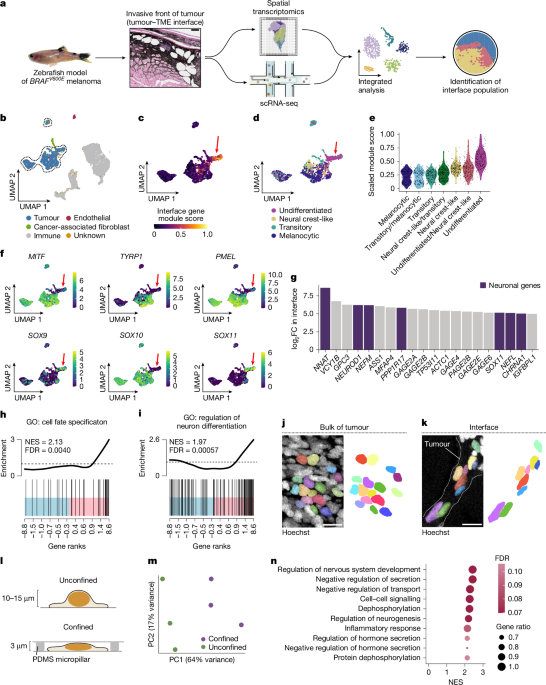
"To study the influence of the local microenvironment on tumour invasion, we applied spatially resolved transcriptomics and single-cell RNA sequencing (scRNA-seq) to a transgenic zebrafish model of BRAFV600E-driven melanoma (Fig. 1a). Tumours from this model frequently invade into adjacent tissues, including the underlying dermis and muscle. Across fish, we found a conserved 'interface' transcriptional cell state that occurs where tumour cells invade into the microenvironment6 (12.1% of zebrafish tumour cells; Fig. 1a)."
Mechanical confinement by the adjacent microenvironment induces stable changes in chromatin architecture that cause melanoma cells to transition from a proliferative to an invasive state. Spatially resolved transcriptomics and single-cell RNA sequencing were applied to a transgenic zebrafish model of BRAFV600E-driven melanoma to study local microenvironment influence on invasion. Tumours in this model frequently invade adjacent dermis and muscle. A conserved 'interface' transcriptional cell state occurs where tumour cells invade into the microenvironment, comprising 12.1% of zebrafish tumour cells. Phenotypic plasticity enables phenotype shifts without additional DNA mutations and may be regulated by microenvironmental cues.
Read at Nature
Unable to calculate read time
Collection
[
|
...
]