"G-protein-coupled receptor (GPCR) signalling occurs through heterotrimeric G proteins, whose selective activation leads to distinct cellular outcomes. Although more than 200 GPCR-G protein complex structures have been determined, these static snapshots provide limited insight into the dynamics of G-protein association and dissociation."
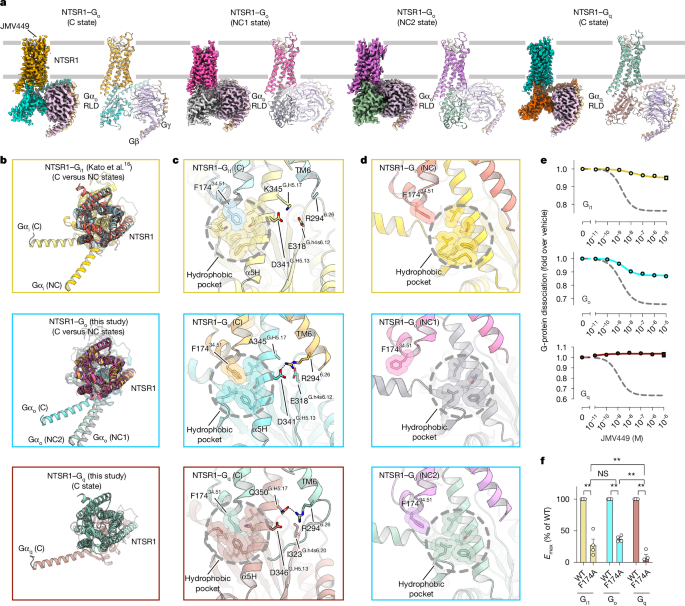
"Time-resolved cryo-electron microscopy analyses of NTSR1-G i visualized G-protein dissociation processes on GDP/GTP binding. Characterization of more than 20 intermediates, complemented by mutational and computational analyses, identifies four key mechanistic features including GDP/GTP-induced G i release from both canonical and non-canonical active conformations with distinct kinetics."
"NTSR1 uses common intracellular rearrangements to recognize different G-protein subtypes and to promote activation of a single subtype. Separation from Gβγ involves stepwise remodelling of the Gα switches I-III."
Cryo-electron microscopy structures reveal how human neurotensin receptor type 1 (NTSR1) interacts with different G-protein subtypes (Go, Gq, and Gi). The receptor's intracellular surface undergoes dynamic rearrangements to accommodate each G-protein variant. Time-resolved cryo-electron microscopy analysis of NTSR1-Gi complexes visualized G-protein dissociation processes triggered by GDP/GTP binding. Analysis of over 20 intermediates identified four key mechanistic features: GDP/GTP induces Gi release from both canonical and non-canonical active conformations with distinct kinetics; NTSR1 uses common intracellular rearrangements to recognize different G-protein subtypes while promoting activation of a single subtype; and Gβγ separation involves stepwise remodeling of Gα switch regions I-III.
#g-protein-coupled-receptors #cryo-electron-microscopy #g-protein-dynamics #ntsr1-signaling #protein-structure
Read at Nature
Unable to calculate read time
Collection
[
|
...
]