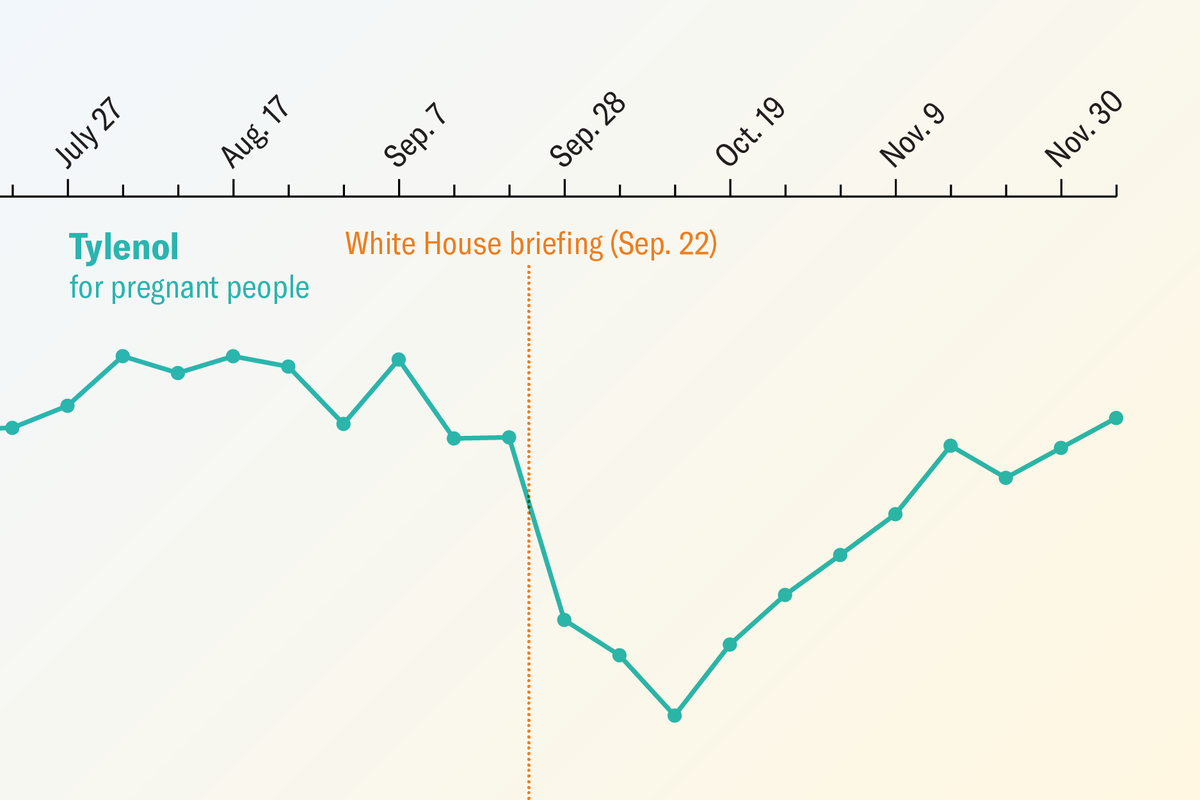
"Trump, along with Secretary of Health and Human Services Robert F. Kennedy, Jr., recommended, without evidence, the use of a medication called leucovorin (folinic acid) for the treatment of autism. In the weeks after Trump's announcement, the number of pregnant patients in emergency rooms who took paracetamol (another name for acetaminophen that is used internationally) decreased by up to 20 percent, according to an analysis published on Thursday in the Lancet."
"The study also found that the number of children aged five to 17 who were prescribed leucovorin as outpatients increased by 71 percent. The researchers included 88,857 pregnant patients and 853,216 nonpregnant patients in the analysis. The findings suggest that the Trump administration's announcement had a notable effect, though it's unclear from the data whether orders for acetaminophen changed because of patients refusing the pain reliever or physicians making a clinical decision."
In September 2025, President Trump announced without scientific evidence that acetaminophen posed increased autism risk and recommended pregnant women avoid it unless necessary. He and HHS Secretary Robert F. Kennedy Jr. promoted leucovorin as an autism treatment despite lacking evidence. Following this announcement, emergency room acetaminophen use among pregnant patients dropped 20%, while leucovorin prescriptions for children aged 5-17 increased 71%. A Lancet study analyzing 88,857 pregnant and 853,216 nonpregnant patients found no change in nonpregnant acetaminophen use, suggesting the decline resulted from both patient refusal and physician decisions. The announcement's impact demonstrates how unsupported health claims can significantly alter medical treatment patterns.
#misinformation-and-health-policy #acetaminophen-and-pregnancy #autism-treatment-claims #medical-decision-making-impact #public-health-consequences
Read at www.scientificamerican.com
Unable to calculate read time
Collection
[
|
...
]