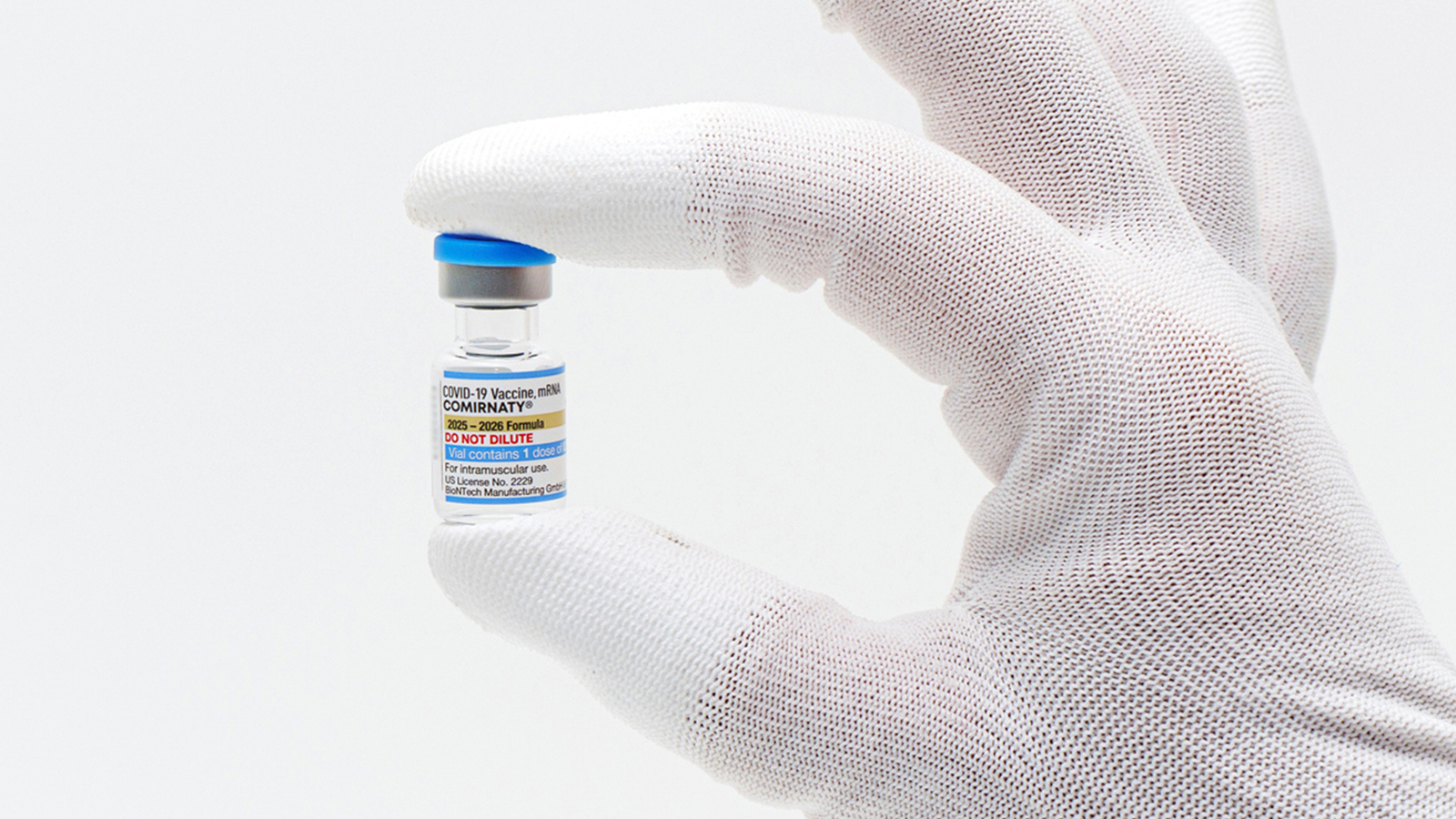
"The new shots from Pfizer, Moderna and Novavax are approved for all seniors. But the Food and Drug Administration narrowed their use for younger adults and children to those with at least one high-risk health condition, such as asthma or obesity. That presents new barriers to access for millions of Americans who'd have to prove their risk -- and millions more who may want to get vaccinated and suddenly no longer qualify."
"Additionally, Pfizer's vaccine will no longer be available for any child underage 5, after the FDA said it was revoking the shot's emergency authorization for that age group. Parents will still be able to seek out shots from rival drugmaker Moderna, the other maker of mRNA vaccines, which has full FDA approval for children as young as 6 months. But the company's Spikevax vaccine is only approved for children with at least one serious health problem."
Updated COVID-19 vaccines from Pfizer, Moderna and Novavax received FDA approval for seniors. Use for younger adults and children was narrowed to persons with at least one high-risk health condition, such as asthma or obesity, creating new access barriers for millions who must prove eligibility. Pfizer's vaccine authorization was revoked for children under 5, while Moderna's Spikevax retains approval for children as young as 6 months but only for those with serious health problems. The revamped vaccines target a newer viral variant and will begin shipping immediately. Implementation and public access will depend on federal advisers, insurers, pharmacies, and state decisions. The policy departs from prior annual-shot recommendations and reflects heightened skepticism about ongoing COVID risks and need for yearly boosters voiced by Health Secretary Robert F. Kennedy Jr. and FDA Commissioner Marty Makary.
Read at ABC7 Los Angeles
Unable to calculate read time
Collection
[
|
...
]